Nobel Prize in Physiology or Medicine 2025
Immune tolerance or the immune system’s brake pedal
Our body’s immune system is vital to our survival. Given the enormous breadth of germs that have the potential to make us ill, which surround us every day – not just in winter – severe infections are strictly speaking rare. Our (fully functioning) immune system acts as an efficient “health police force” protecting us from pathogenic microorganisms and even abnormal (cancerous) cells. It is a complex interplay of different immune cells from our innate and adaptive immune system that affords us this protection. They detect and eliminate foreign and abnormal cells and they also build up an antibody-based immune protection.
Nevertheless, as efficient and vital to survival as this cellular health police force may be, moderation – as with so many things in life – is key. Every immune system has to be kept in balance at all costs. It must reliably be able to distinguish between friend and foe, that’s to say between the body’s own cells and foreign cells, and to quickly downregulate after successful deployment. Problems arise when this is not the case, because an off-balance immune system is a serious threat to our body: dangerous overreactions of the immune system can even send the body into life-threatening shock, in autoimmune diseases, immune cells attack and destroy the body’s own structures. The difficult yet essential task of keeping our body’s defences in check lies with another cellular guardian, as discovered and characterised by Shimon Sakaguchi, Mary E. Brunkow and Fred Ramsdell, earning them the Nobel Prize in Physiology or Medicine in late 2025. What did they do?
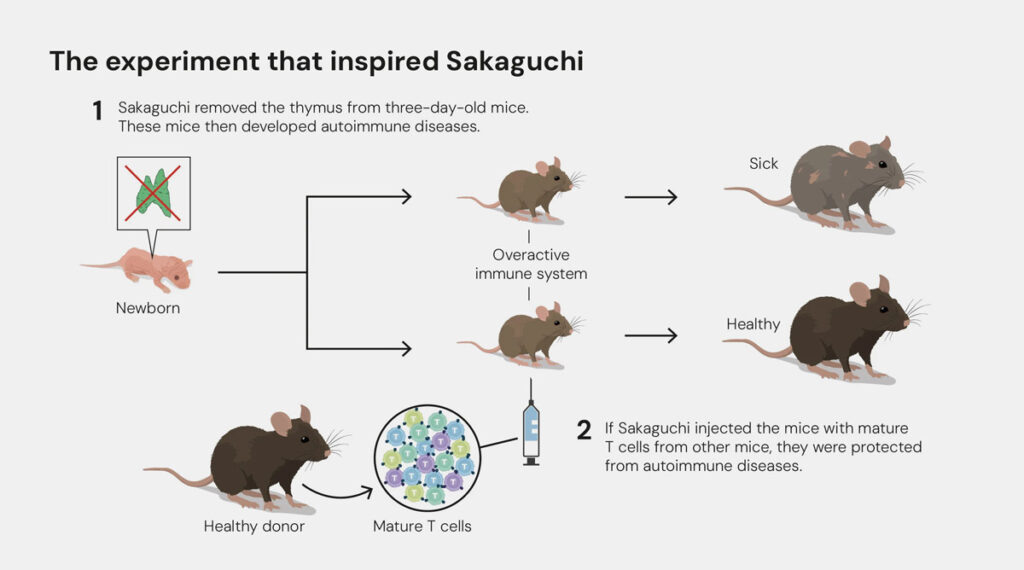
In 1995 Sakaguchi discovered a new class of immune cells, known as regulatory T cells – Treg or Treg for short. Strictly speaking, scientists were already aware of these cells in the 1970s – albeit under the name “suppressor cells” back then. However, methods at that time could not reliably distinguish these from other T cells. And this is precisely what Sakaguchi ultimately achieved through his pioneering work. The answer lay in sophisticated analytics and a special surface marker developed only by the Tregs within the large group of T-lymphocytes. This marker, called CD25, identifies the alpha chain of the Interleukin-2 receptor.
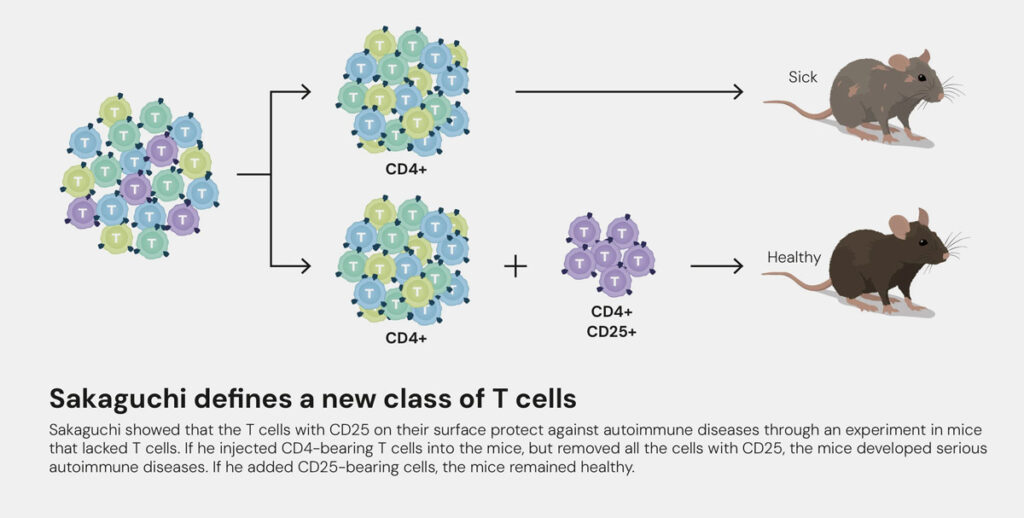
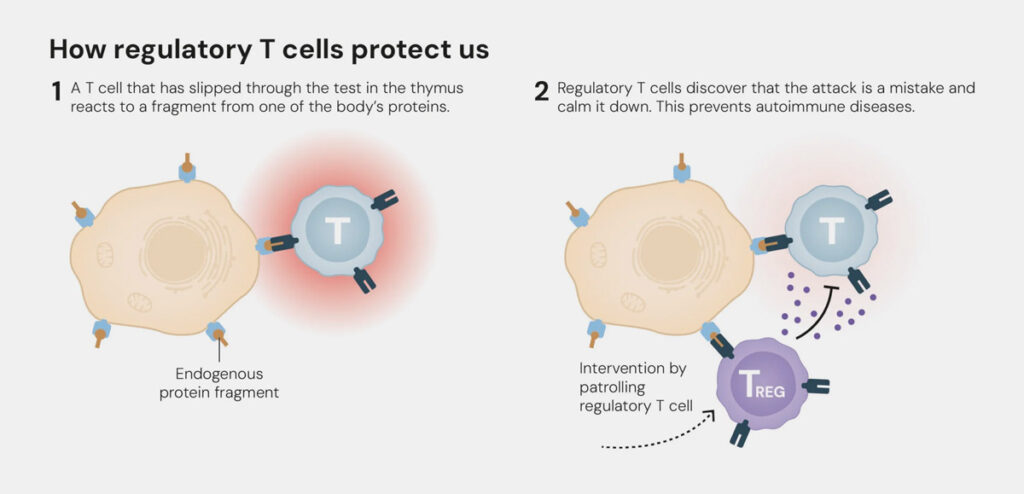
It became apparent that the Tregs act as “immune system security guards”, preventing the body’s defence system from attacking its own organism. As such, they have an immunoregulatory function. This gives rise to a state that researchers call immune tolerance. Until this was discovered by Sakaguchi, the prevailing scientific opinion was that immune tolerance was primarily regulated centrally in the thymus. T lymphocytes are produced in the bone marrow and mature in the thymus, where they are programmed for their subsequent tasks. Immune cells that mistakenly target the body’s own structures are thereby “weeded out”, i.e. they are driven to programmed suicide, in a process called apoptosis. But every system is fallible. Sakaguchi discovered the regulatory T cells had the additional control capability required to suppress overactive immune responses or those that turn against the body’s own structures. And all this happens right at the heart of proceedings – in our blood, in the lymphatic system and in the tissue.
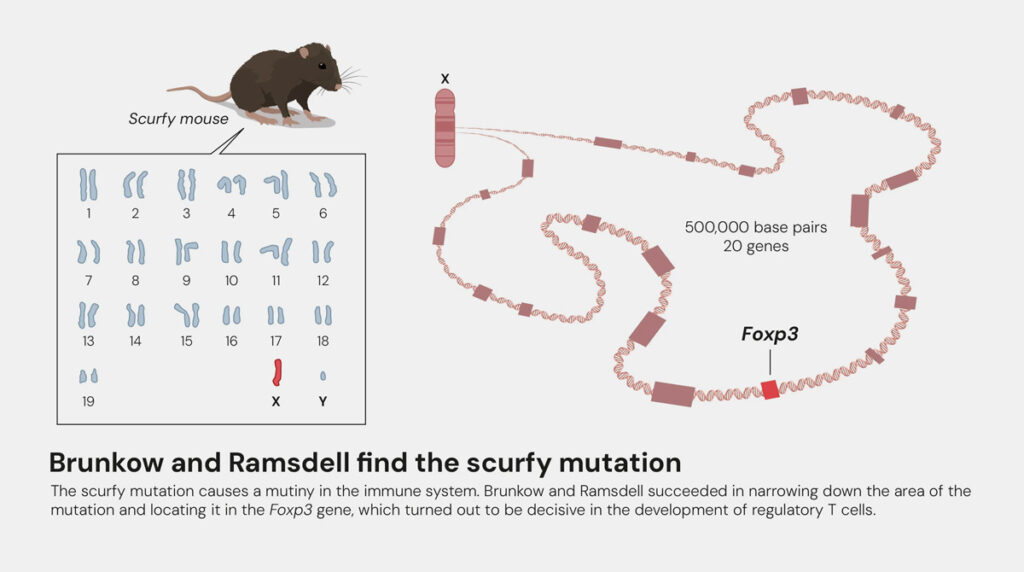
Meanwhile, in 2001 Mary E. Brunkow and Fred Ramsdell happened upon the genetic basis of a severe autoimmune disease: immunodysregulation polyendocrinopathy enteropathy X-linked syndrome (IPEX syndrome). The scientists identified the cause to be a mutation of the FOXP3 gene, which can also be found in so-called scurfy mice with autoimmune disease.
Two years later, Ramsdell and Sakaguchi successfully linked these two discoveries together, by proving that the FOXP3 protein controls the development of regulatory T cells, acting as a transcription factor, or to a certain extent as an “on-/off switch”. This established the connection between regulatory T cells, autoimmune diseases and FOXP3 and the concept of peripheral immune tolerance was born.
“We now have a better understanding of how the immune system works and why we don’t all develop a severe autoimmune disease,” reasons the Nobel committee upon announcing their decision to the prize winners.
But how exactly do the Tregs manage to put the brakes on our immune system without weakening its overall powers of defence? And how can these fundamental findings by this year’s Nobel prize winners be expanded and utilised?
How do regulatory T cells regulate the immune system – and who or what is regulating them?
One way they do this is by slowing down the division activities of T helper cells while at the same time restricting their production of immune messengers, by blocking key transcription factors in the helper cells. In this way, Tregs suppress inflammatory processes and actively contribute to tissue regeneration by releasing cytokines.
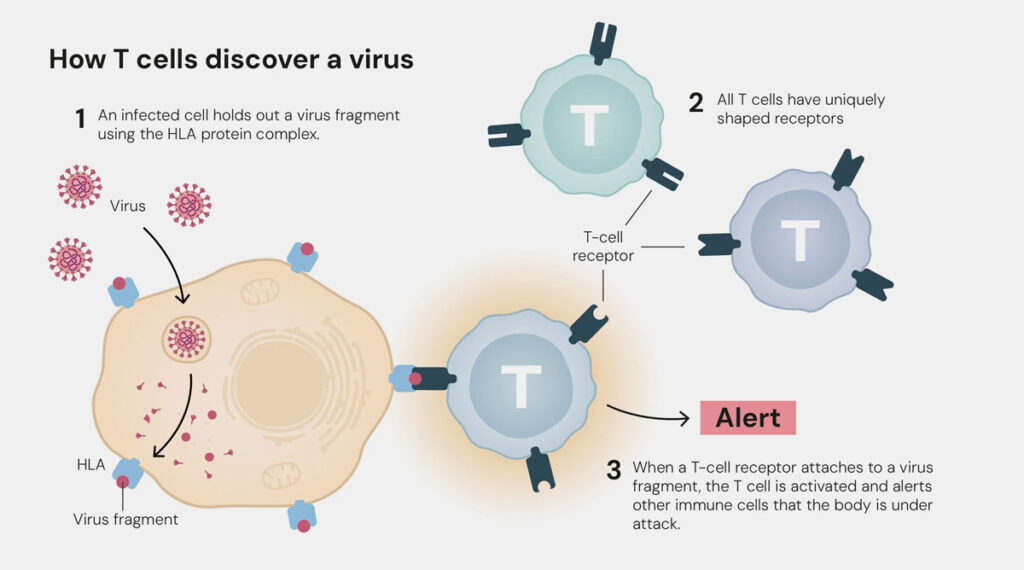
T helper cells are activated by means of antigen binding (e.g. virus surface proteins) and then release cytokines. These messengers activate immune cells (e.g., B cells and cytotoxic T cells) and, in turn, a specific immune response to pathogens and cancer cells. T helper cells are an important element of adaptive immunity and serve as key players in the body’s fight against infections, autoimmune diseases, allergies and cancer.
However, if these immune system brakes were applied permanently, our defences would be weakened. It is precisely this effect that is exploited by abnormal cells in tumours, by gathering regulatory T cells around themselves and, in doing so, limiting the body’s healing immune response to the tumour. Therefore, the regulators must themselves be regulated.
The Tregs are activated on an antigen-specific basis and the cells multiply faster than normal T cells, quickly triggering a high level of activity. They are deactivated promptly and automatically: Tregs are extremely unstable, meaning they die comparatively quickly through apoptosis.
Enormous potential for application albeit with some obstacles
Not only does the work of the three Nobel prize winners provide fundamental basic knowledge, but the Tregs themselves also present enormous therapeutic potential. As an “active ingredient” for the treatment of conditions associated with an overactive or misguided immune system, Tregs could help us to better control autoimmune diseases, allergies or even rejection responses following organ transplants. If, by contrast, we weaken the effect of the Tregs – severing the “immunity brakes” so to speak – this could be beneficial to cancer treatment. CAR T cell therapy, which has emerged in the past few years, could also benefit from Tregs if this new form of treatment were expanded to diseases with an overactive immune system, such as certain types of hepatitis or neurodegenerative diseases, type 1 diabetes or multiple sclerosis.
However, long after their discovery, Tregs are still limited by one general issue when it comes to exploiting their therapeutic potential. The very characteristic that is vital to preventing Tregs from permanently weakening the body’s immune system also presents a huge obstacle for further research, optimisation and clinical application: namely, their instability. Not only does this present a challenge when it comes to extracting, purifying and expanding the cells in sufficient quantities for therapeutic purposes, but the Tregs also have a tendency to revert to “normal” T cells in laboratory conditions. Countless clinical studies on Treg-based therapies, such as for type 1 diabetes, Lupus erithematodes, rheumatoid arthritis or Hashimoto’s thyroiditis, have already been outlined, but as yet none has got beyond clinical phases I or II.
That could conceivably change: of all the study groups working on the subject, it is the group led by freshly decorated Nobel prize winner Sakaguchi that has now published two scientific papers, in which researchers describe the protocols for the stable production of Tregs from mice and humans [1] as well as their application for the autoimmune disease pemphigus vulgaris. Sakaguchi and his team also developed a protocol for the pharmacological epigenetic reprogramming of T cells and used this to transform conventional (stable) CD4+ T helper cells into (also stable) Tregs. It is hoped that these approaches can be established for a broader field of application. Then, Tregs could to some extent (boosted along by the Nobel prize) finally climb into the realms of clinical application and realise its full potential. Goodness knows they deserve it.
Cited sources:
[1] N. Mikami et al., 2025, Science Translational Medicine DOI: 10.1126/scitranslmed.adr6049
[2] M. Mukai et al., 2025, Science Translational Medicine DOI: 10.1126/scitranslmed.adq9913
Other sources:
Pressemitteilung: Nobelpreis für Physiologie oder Medizin 2025 – NobelPrize.org
Scientific background to the Nobel Prize in Physiology or Medicine 2025
Immuntoleranz im Fokus: Medizin-Nobelpreis 2025
Medizin-Nobelpreis 2025 für Erforschung der Immunregulation – Spektrum der Wissenschaft
Medizin-Nobelpreis 2025 für Forschung zur Immuntoleranz
Wie regulatorische T-Zellen das Immunsystem kontrollieren
Potente Sensibelchen in der Klinik | PZ – Pharmazeutische Zeitung
239 Regulatorische T-Zellen (Treg) als Basismarker zum Monitoring immunmodulierender Maßnahmen
Selbstkontrolle des Immunsystems dämpft Krebsabwehr – Deutsches Krebsforschungszentrum
Wie regulatorische T-Zellen reguliert werden – MT-Dialog
Clinical studies:
Medizinische Hochschule Hannover : Bio-Navi gegen fehlgeleitetes Immunsystem
Fehlgeleitete Immunreaktionen: Regulatorische CAR-T-Zellen entwickelt – Biermann Medizin