
Nobel Prize in Chemistry 2025
Porous and prize-worthy: how MOFs are changing the world
On 8th October 2025, the Royal Swedish Academy of Sciences awarded three researchers the scientific equivalent of an Olympic medal: Susumu Kitagawa, Richard Robson and Omar M. Yaghi received the Nobel Prize in Chemistry for their research on metal-organic frameworks, or MOFs for short. These versatile structures are often simplistically described as molecular sponges because they are characterised by their porous structure, which allows them to absorb gas molecules or other substances, for example. This function gives MOFs enormous application potential and could solve some of the most pressing environmental problems of our time.
Enchanting capacity for guest molecules
Metal-organic framework compounds consist of metal ions that are linked together by organic molecules. This creates a regular framework with cavities that offer plenty of space to accommodate other molecules. “A bit like Hermione’s handbag from Harry Potter – it fits more in it than you think,” explains Prof. Heiner Linke, Member of the Nobel Committee by way of comparison.
This analogy from the world of magic comes quite close to reality: even in the small volume of a sugar cube, a MOF can offer the surface area of a football field. The cavities of the framework connections can be specifically adapted by selecting the appropriate metallic and organic precursors. Depending on the combination of building blocks, MOFs can absorb and store certain substances.
From wooden ball to Nobel Prize discovery
The history of MOFs began in 1974 with some everyday lecture preparation. Robson was teaching at the University of Melbourne and wanted to build molecular models for his students. As he assembled the molecules from rods and wooden balls, he realised how much information was contained in the arrangement of the drill holes alone – the bond angles and number of bonds ultimately determine the shape and structure of the molecules. This gave the researcher an idea: could such bonding properties also be used to link entire molecules together instead of just atoms?
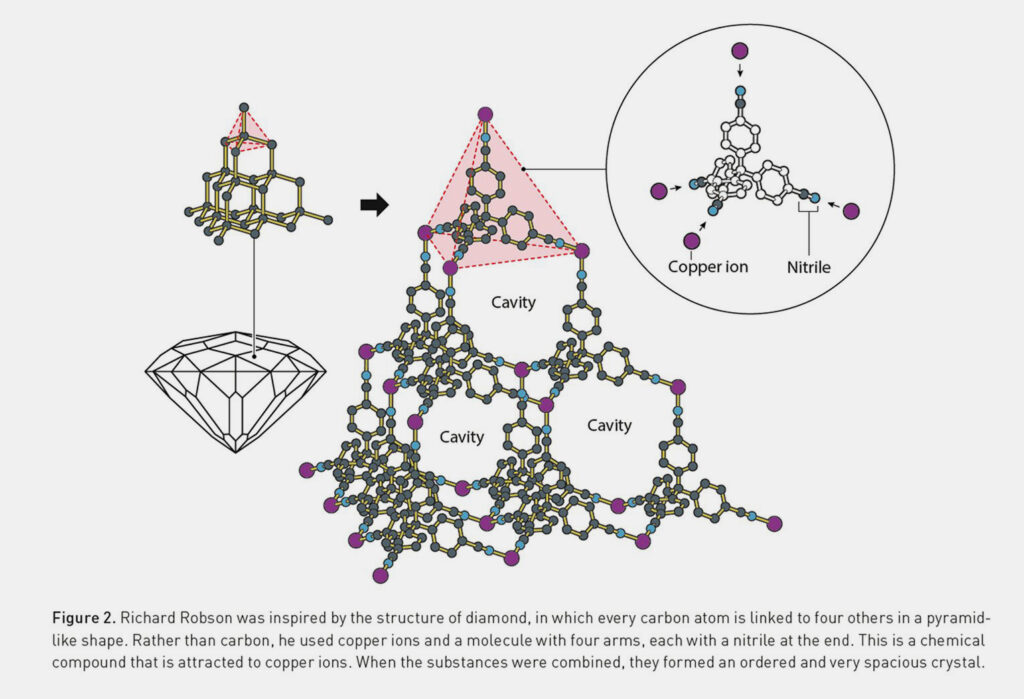
More than a decade passed before Robson put his idea into action. In 1989, he combined positively charged copper ions with a four-armed molecule. This did not result in chaotic structures, but rather an orderly molecular lattice comparable to the regular crystalline structure of carbon atoms in a diamond. However, while the latter is very compact, Robson’s compound contained a multitude of large cavities. This laid the foundation for MOF chemistry.
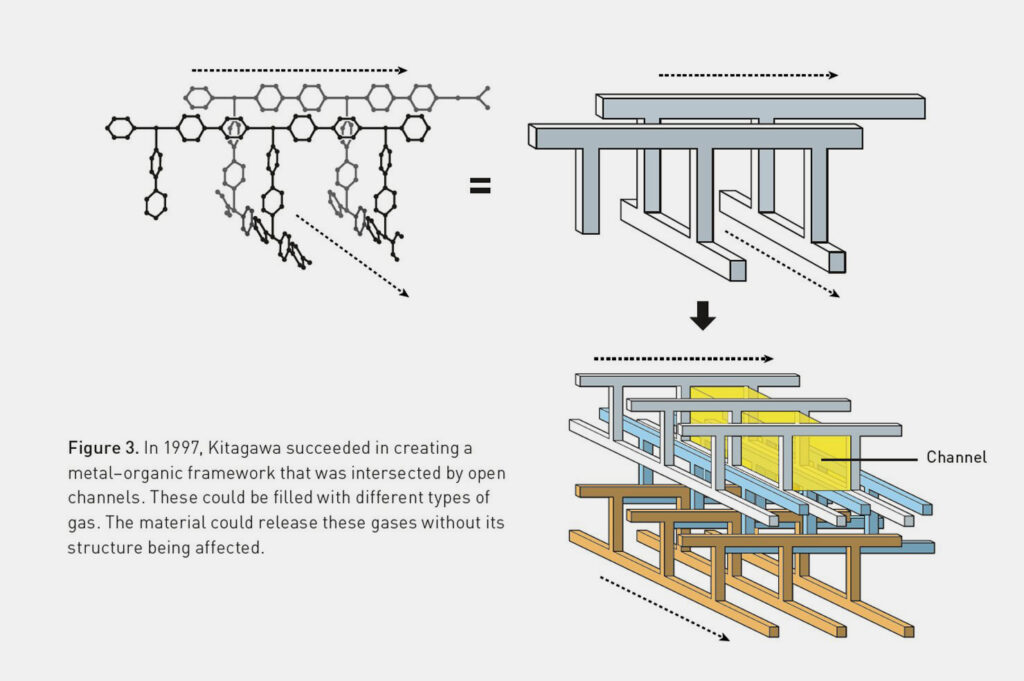
Kitagawa and his research team achieved a decisive breakthrough in the field of metal-organic framework compounds in 1997. They created stable, three-dimensional MOFs that were criss-crossed by open channels. In experiments, the scientists showed that the cavities in the compounds could be filled with gases. This allowed methane, nitrogen and oxygen to be absorbed and released without changing the shape of the MOF crystal lattice.
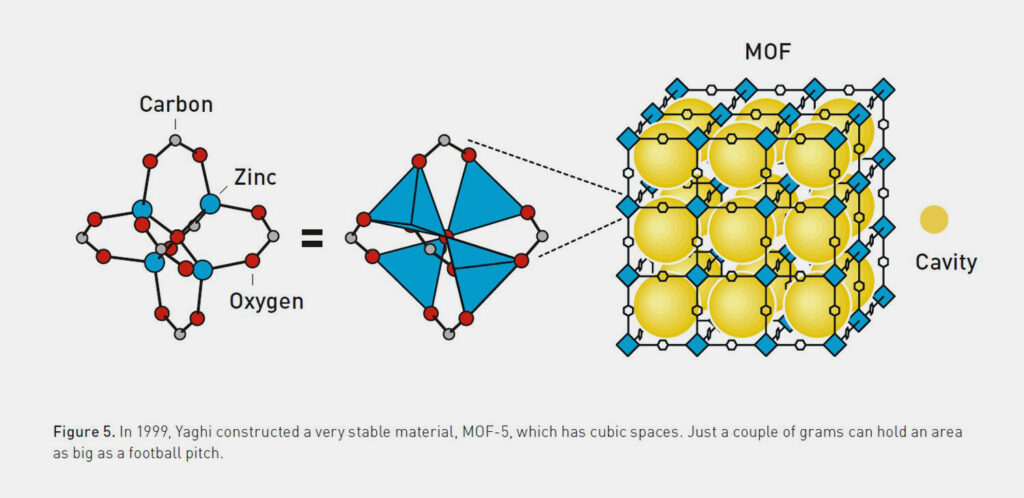
Yaghi made further progress with his research. In 1999, he synthesised “MOF-5” – an exceptionally absorbent and stable material. The highly porous compound proved to be extremely robust and could be heated to around 300 °C without collapsing. What was truly remarkable, however, was the enormous specific surface area of the new compound: just a few grams of MOF-5 offer a surface area as large as a football field. This allows it to adsorb more gas than typically used zeolite materials.
Yaghi also carried out research on the reticular synthesis of the compounds. This allows new MOFs to be developed from a known basic structure with little effort. Only the organic connecting pieces are replaced, while the metal ions in the lattice remain unchanged. This results in structurally related MOFs with different properties.

Extracting water from desert air and other areas of application
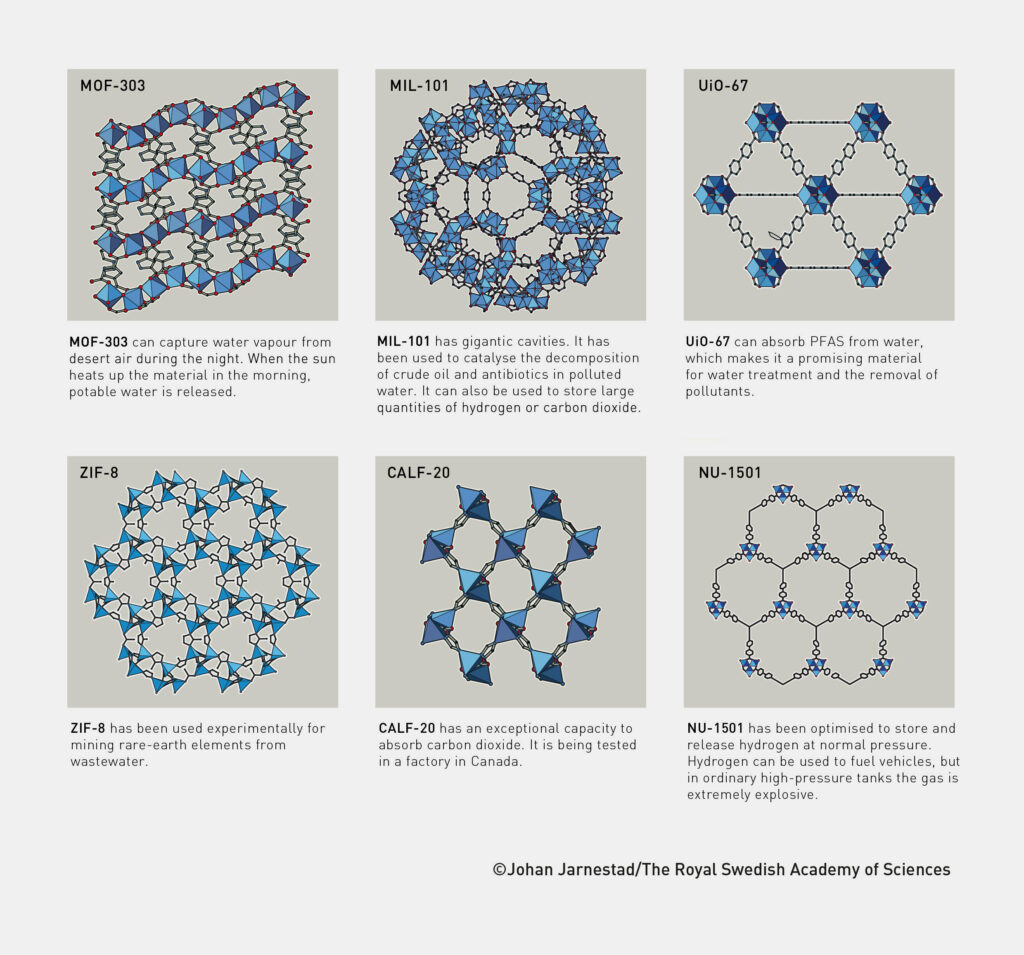
Today, there are tens of thousands of different MOFs. They can be used to remove carbon dioxide from the air or pollutants from water. But other applications are also being researched, such as extracting drinking water from dry desert air. Yaghi’s research group has already demonstrated how this works: in the Arizona desert, their MOF-303 captures water vapour from the air at night. When the sun warms the material in the morning, the water is released and can be extracted as drinking water.
Further application examples for MOFs:
- removing PFAS from water with UiO-67
- recovering rare earth elements from wastewater with ZIF-8
- storing carbon dioxide with CALF-20
- storing hydrogen at normal pressure with NU-1501
- purifying water (through catalytic decomposition of crude oil and antibiotic residues) with MIL-101
A new construction kit for chemistry
MOFs have now moved beyond the pure research stage, and many companies are already investing in their mass production and marketing. For example, MOFs are used in the electrical industry to bind some of the toxic gases used in the manufacture of semiconductors. And since the variance of MOFs is essentially unlimited, many more applications are conceivable in the future. Through the development and characterisation of metal-organic frameworks, Kitagawa, Robson and Yaghi have opened a new door for chemists around the world and created opportunities to solve some of the greatest challenges of our time. This has now been recognised by the Nobel Prize Committee.
The three researchers will share the prize money, which is equivalent to just under one million euros. Each of the three researchers can fully claim the highly prestigious title of Nobel Prize winner.
The scientific background
MOFs are based on the coordination chemistry between metal ions and organic compound molecules, known as linkers. In his pioneering work, Robson used Cu⁺ ions with 4′,4″,4‴,4⁗-tetracyanotetraphenylmethane as a tetrahedral linker, creating a diamond-like framework structure with large cavities.
Yaghi supplemented MOF chemistry with the concept of secondary building units (SBU), inspired by zeolite chemistry. His MOF-5 is based on Zn₄O carboxylate clusters as SBUs, which are linked with 1,4-benzoldicarboxylate to form a highly stable structure. MOF-5 achieves a pore volume of 0.6 cm³/ml and Langmuir surfaces of 2900 m²/g. By comparison, zeolites typically have a surface area of several hundred square metres per gram. Newer MOFs such as MOF-210 even significantly exceed these values and achieve specific surface areas of over 10,000 m²/g (Langmuir).
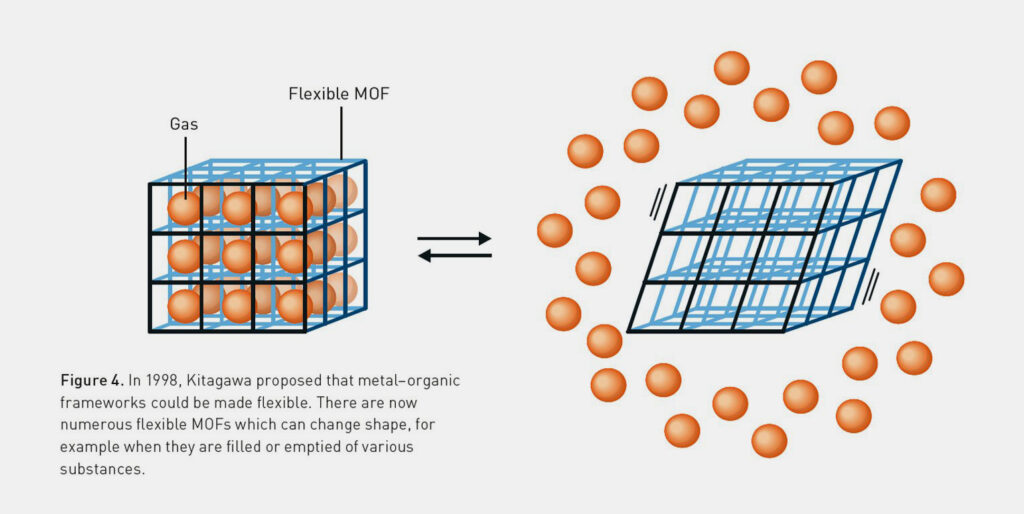
Finally, Kitagawa classified MOFs into three generations: unstable (first), stable (second) and dynamic structures (third generation). He synthesised a tongue-and-groove framework from Co2⁺, 4,4′-bipyridine and nitrate. This compound changes its morphology in response to external stimuli such as temperature and pressure.
Images: ©Johan Jarnestad/The Royal Swedish Academy of Sciences
Sources:
https://www.nobelprize.org/prizes/chemistry/2025/popular-information
https://www.nobelprize.org/prizes/chemistry/2025/press-release
The Nobel Committee for Chemistry: Metal-Organic Frameworks, Scientific Background to the Nobel Prize in Chemistry 2025, 8 October 2025 https://www.nobelprize.org/uploads/2025/10/advanced-chemistryprize2025.pdf

