Outwitted cancer cells
How medicine is optimising cancer treatments through checkpoint inhibitors
Karin Hollricher
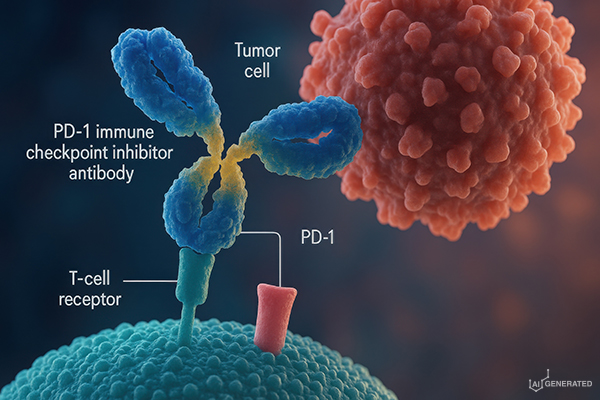
Checkpoint molecules regulate the ability of the immune system to start or end an immune response as required. Cancer cells are often able to manipulate these regulatory mechanisms in such a way that the immune system fails to combat the malignant cells, for example via the three receptors, CTLA-4, PD-1 and LAG-3, located on the surface of T cells. These receptors inhibit the activity of their T cells when specific ligands from cancer cells bind to them. Checkpoint inhibitors – molecules that can override this insidious strategy – are now an integral part of cancer treatment.
In 2011, the antibody Ipilimumab became the first checkpoint inhibitor to be introduced into clinical practice. When it binds to the CTLA-4 receptor, it prevents the recognition of the B7 ligand, which cancer cells produce in increased quantities. Antibodies that block the binding of the ligand PD-L1 to its receptor PD-1 (Programmed Cell Death 1) work on the same principle. Protected in this way, cytotoxic cells can become active and ensure the destruction of the cancer cells. Furthermore, the immune response creates an immune memory that can combat tumour cells permanently.
“We need to address the intricacies of other immunological and non-immunological signalling pathways that cumulatively influence anti-tumour immunity,”
says Nobel laureate James Allison, who not only discovered the T-cell receptor but also recognised the function and use of CTLA-4 [5].
Many types of tumours respond to checkpoint inhibitors, which is reflected in longer progression-free survival and also longer overall survival. Both are crucial metrics for evaluating oncological therapeutics. Not all, but many patients can even live for years without the disease progressing. These drugs achieved a real breakthrough in the fight against malignant melanoma [1]. The ten-year survival rate for people with stage IV melanoma – a highly advanced form of the disease – was less than 10% until 2015, when treatment with PD-1/PD-L1 and/or CTLA-4 inhibitors was approved. This survival rate is now thought to be approaching 45% – precise data on this is likely to be available soon [2, 3].
In 2015, former US President Jimmy Carter was diagnosed with metastatic melanoma. Thanks to a checkpoint inhibitor, he was able to overcome this disease, which would certainly have claimed his life within a short time in the past. He lived to the age of 100 and passed away at the end of 2024 – but not from this cancer.
The receptor LAG-3 is the third molecule in the checkpoint family. LAG stands for “Lymphocyte Activation Gene” – which adequately describes its function. Its actual ligands are molecules belonging to the MHC class II family. Relatlimab counteracts these immune brakes; it is the only antibody and checkpoint inhibitor approved to date that targets this checkpoint.
Currently, there are several antibodies authorised in Europe against PD-1 and two against CTLA-4, intended for use in combating numerous oncological diseases ranging from Hodgkin’s lymphoma to lung tumours and ovarian cancer. In addition, antibodies are available that do not bind to the receptor itself, but instead neutralise the PD-L1 ligand. Further active substances are undergoing clinical trials and the approval process, which is expected to both broaden the range of indications and improve the overall efficacy.
Although this form of immunotherapy has established itself as the fourth pillar of cancer treatment alongside surgery, radiotherapy and chemotherapy, it is by no means a miracle cure. This is likely due to the fact that the human immune system is subject to highly complex regulation. For example, the molecule galectin-9 appears to be a highly effective additional inhibitor of immune competence by binding to the T-cell receptor TIM. At least, that is what the team led by Martina Seiffert at the German Cancer Research Centre in Heidelberg (DKFZ) suspects, based on data obtained through a comprehensive single-cell analysis of the immune cells of patients with chronic lymphocytic leukaemia (CLL) [4]. To date, CLL has not responded to checkpoint inhibitors. Research of this kind could pave the way for new immunotherapies for patients whose disease is resistant to existing drugs. The development of immunomodulatory agents is therefore far from over.
Sources:
[1] J. Wolchok et al., 2025, N. Engl. J. Med. 392, 11-22
[2] www.vfa.de/de/arzneimittel-forschung/weltkrebstag-malignes-melanom
[3] S. Bhatia et al., 2009, Oncology 23, 488-496
[4] L. Llaó-Cid et al., 2025, Nat. Commun. 16, 7271
[5] P. Sharma et al., 2023, Cell 186, 1652-1669

